|
8/10/2023 0 Comments Asymmetric carbon atom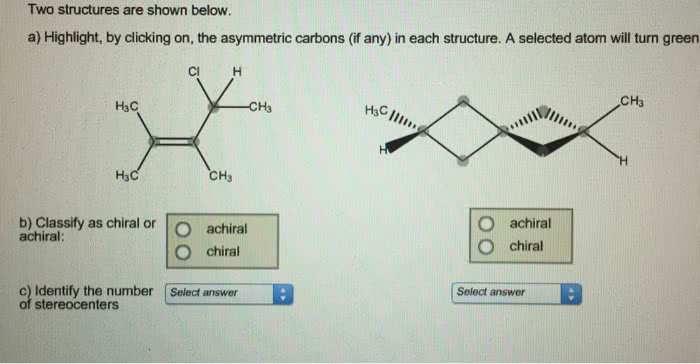
Planar chirality may also provide for chirality without having an actual chiral center present.Ĭonfiguration is defined as the arrangement of atoms around a stereocenter. Cyclic compounds with chiral centers may not exhibit chirality due to the presence of a two-fold rotation axis. 
Īdditionally, certain configurations may not exist due to steric reasons. Resultantly, a meso compound will reduce the number of stereoisomers to below the hypothetical 2 n amount due to symmetry. Since a meso compound is superposable on its mirror image, the two "stereoisomers" are actually identical. However, the stereoisomers produced may also give a meso compound, which is an achiral compound that is superposable on its mirror image the presence of a meso compound will reduce the number of possible stereoisomers. Enantiomers and diastereomers will produce individual stereoisomers that contribute to the total number of possible stereoisomers. Enantiomers and diastereomers are produced due to differing stereochemical configurations of molecules containing the same composition and connectivity (bonding) the molecules must have multiple (two or more) stereocenters to be classified as enantiomers or diastereomers. The stereoisomers produced by the presence of multiple stereocenters can be defined as enantiomers (non-superposable mirror images) and diastereomers (non-superposable, non-identical, non-mirror image molecules). However, this is an upper bound because molecules with symmetry frequently have fewer stereoisomers. 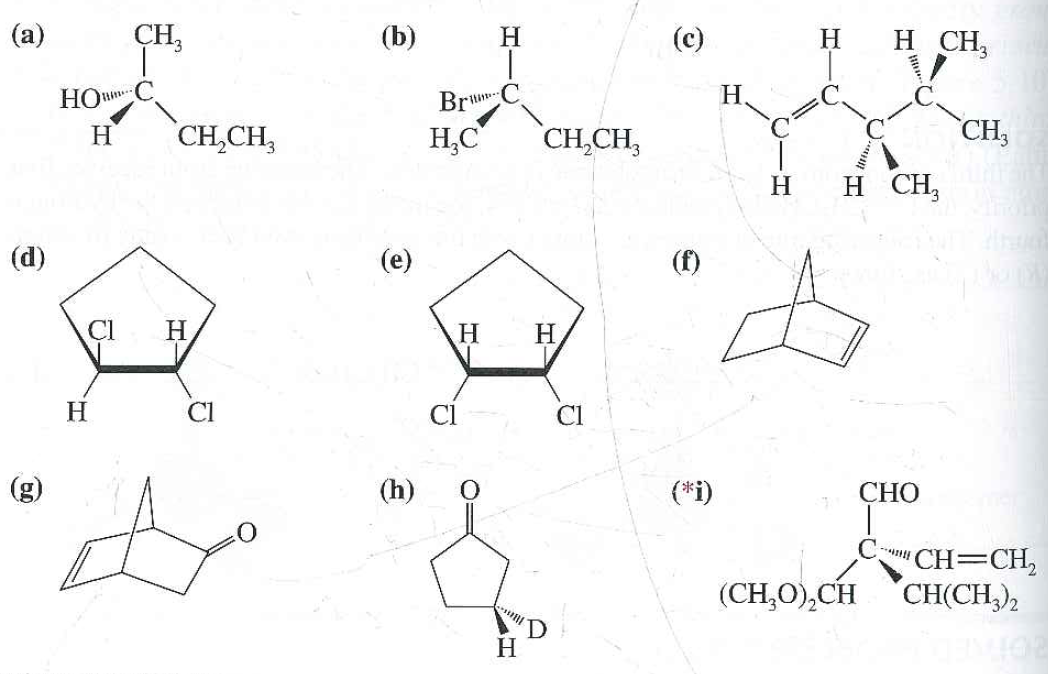
In compounds whose stereoisomerism is due to tetrahedral (sp 3) stereogenic centers, the total number of hypothetically possible stereoisomers will not exceed 2 n, where n is the number of tetrahedral stereocenters. A molecule having multiple stereocenters will produce many possible stereoisomers. Stereoisomers are compounds that are identical in composition and connectivity but have a different spatial arrangement of atoms around the central atom. Since stereocenters can exist on achiral molecules, stereocenters can have either sp 3 or sp 2 hybridization. A stereocenter can have either four different attachment groups, or three different attachment groups where one group is connected by a double bond. They are defined as a location (point) within a molecule, rather than a particular atom, in which the interchanging of two groups creates a stereoisomer. Stereocenters can exist on chiral or achiral molecules. Ĭhirality centers are a type of stereocenter with four different substituent groups chirality centers are a specific subset of stereocenters because they can only have sp 3 hybridization, meaning that they can only have single bonds. The number of hypothetical stereoisomers can be predicted by using 2 n, with n being the number of tetrahedral stereocenters however, exceptions such as meso compounds can reduce the prediction to below the expected 2 n. Stereocenters can exist on chiral or achiral molecules stereocenters can contain single bonds or double bonds. Stereocenters are also referred to as stereogenic centers.Ī stereocenter is geometrically defined as a point (location) in a molecule a stereocenter is usually but not always a specific atom, often carbon. In stereochemistry, a stereocenter of a molecule is an atom (center), axis or plane that is the focus of stereoisomerism that is, when having at least three different groups bound to the stereocenter, interchanging any two different groups creates a new stereoisomer. Two enantiomers of a generic amino acid at the stereocenter
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
